Stem Cell Therapy & Clinical Trials to treat Vision Loss
You might have heard about stem cell therapy in the news and wondered how it works or if it could help you or someone you know who is living with vision loss. To help answer your questions, we’ve put together some information about stem cells and how stem cell therapies are being tested as treatments for different blinding eye diseases.
What is stem cell therapy?
Stem cell therapy is used to repair or replace damaged cells or tissues. Researchers are studying if stem cell therapy can treat many different kinds of eye disease, including age-related macular degeneration (AMD), inherited retinal diseases (IRDs), glaucoma, and corneal diseases.
What are stem cells?
Stem cells are special because they can make many different cell types such as muscle cells or brain cells or retinal cells (the cells that sense and transmit light signals). Other adult cells can’t do this. For example, a skin cell only makes new skin cells and a liver cell only makes new liver cells. In addition, many nerve cells, including retinal cells, are not able to make any new cells at all so they cannot be easily replaced if they are damaged.
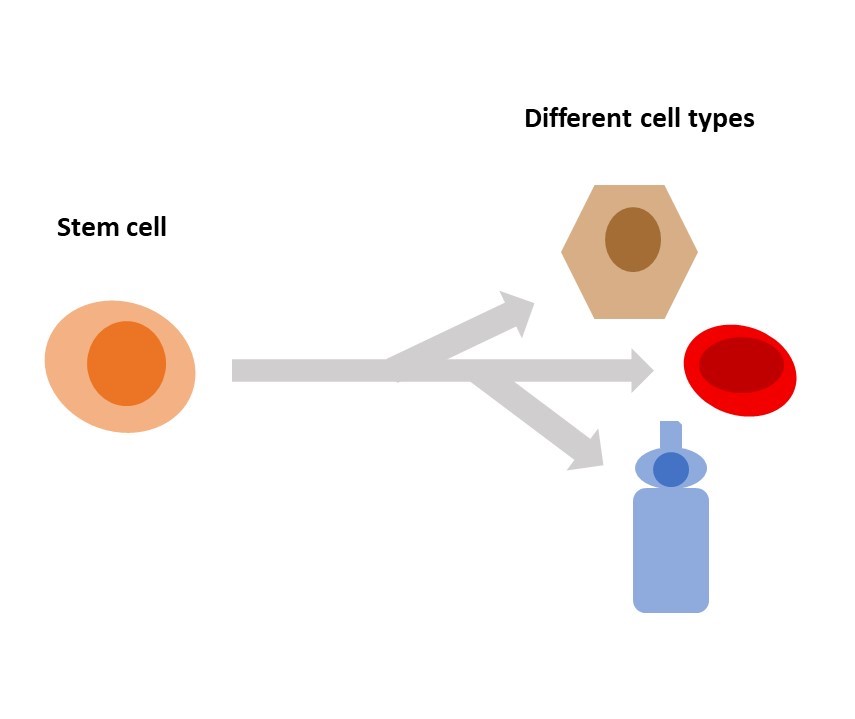
Three main types of stem cells are used in vision research:
Embryonic stem cells (ESCs)
Embryonic stem cells (ESCs) are pluripotent. This means they can become any type of cell in the body. Because of this ability, ESCs have been very important in stem cell therapy research.
Induced pluripotent stem cells (iPSCs)
Induced pluripotent stem cells (iPSCs) are adult cells that have been changed to act like ESCs. About 15 years ago, scientists discovered that they could change the genes of an adult cell such as a skin or a blood cell and turn it into an iPSC. iPSCs can make all cell types and because they can be made in the laboratory there is an unlimited supply of them. iPSCs are now at the forefront of stem cell research.
Adult stem cells
Adult stem cells are found in small numbers in many tissues and can turn into a few but not all types of cells. Some adult stem cells, like bone marrow stem cells, which make blood and immune cells, or corneal limbal stem cells, help tissues heal and regenerate. Adult stem cells exist in the retina and are called retinal progenitor cells. In the body, retinal progenitor cells don’t normally make new retinal cells even if there is damage, but scientists have been able to encourage them to make new retinal cells in the laboratory. Adult stem cells are being studied as treatments for blinding eye diseases, but their use is more limited compared to ESCs or iPSCs.
Stem Cell Therapy for Vision Loss
There is a lot of excitement about stem cell therapy as a treatment for vision loss because a single stem cell therapy may be able to treat different eye diseases. Stem cell therapy could also be useful for individuals whose disease is very advanced and there is a lot of cell damage. In contrast, in gene therapy, which is gene specific, there have to be enough healthy cells left in the eye for the treatment to be successful. Learn more about gene therapy for vision loss.
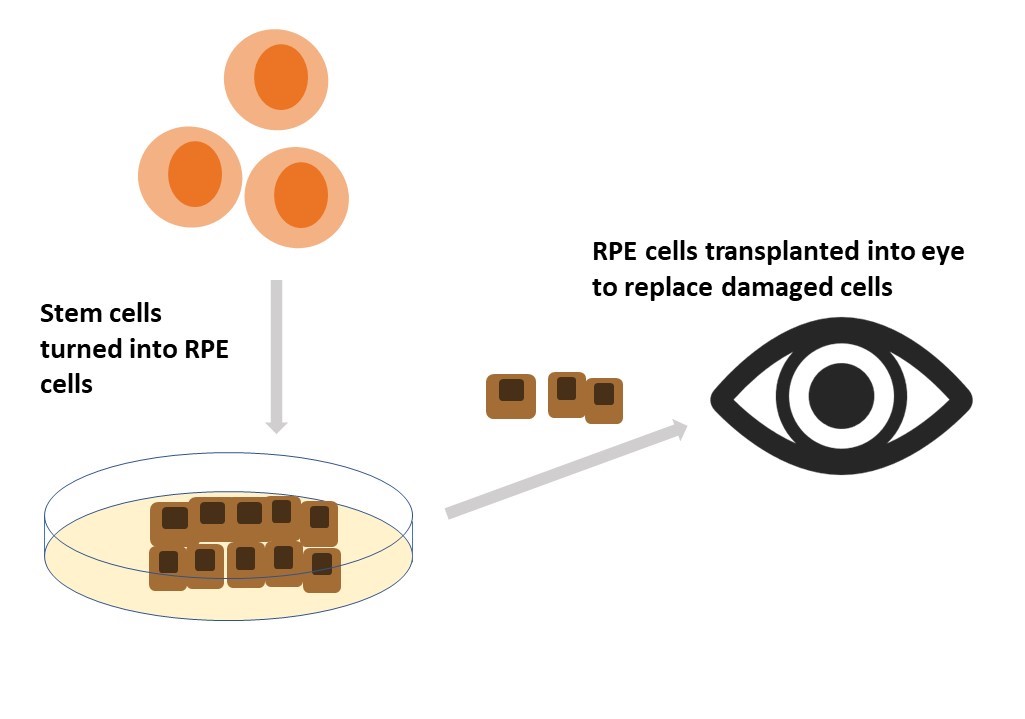
Examples of how stem cells are being used and tested as treatments for vision loss:
- Corneal limbal stem cells from a donor or the patient’s other eye have been used as a treatment for some types of corneal damage or disease.
- Retinal degeneration in diseases like AMD, retinitis pigmentosa or choroideremia can be caused by different factors or genes but ultimately it results in the death of the light sensing photoreceptor cells as well as their support cells, retinal pigment epithelial (RPE) cells. Scientists have had the most success making and transplanting RPE cells and clinical trials are testing if RPE cells, made from ESCs or iPSCs can improve vision for individuals with retinitis pigmentosa, Stargardt disease, or dry AMD.
- There are currently a few clinical trials testing if adult stem cells such as retinal progenitor cells could be a treatment for retinitis pigmentosa. Interestingly, early results show that the main effect of the progenitor cells is protective (helping existing retinal cells survive) but not regenerative (developing into new retinal cells).
- Laboratories around the world are also studying many different types of stem cell therapy for a number of eye diseases. For example, researchers are studying if stem cells can be used to replace damaged cells in the optic nerve for glaucoma or are trying to combine stem cell therapy with other innovative treatments like gene therapy for wet AMD.
Stem Cell Therapy Clinical Trials
Visit our Clinical Trials page to find information about stem cell clinical trials happening around the world. If you are interested in participating in a trial, talk to your eye doctor.
Caution: Be aware of something called stem cell tourism. This is direct-to-consumer marketing by dangerous, unapproved stem cell clinics. These businesses claim that they can treat a wide range of diseases, including blinding eye diseases. However, these are often untested therapies and some patients have lost their eyesight after receiving these treatments. You should never pay money to get an unproven treatment.
For more information about clinical trials, contact our Health Information Line at 1.888.626.2995 or by email at healthinfo@fightingblindness.ca
This resource page is part of FBC’s “Clinical Trials Corner Series”.
LEARN MORE
FBC is proudly funding cutting edge research that is driving the development of new stem cell therapies. Learn more about FBC funded research.
Join the Fight!
Learn how your support is helping to bring a future without blindness into focus! Be the first to learn about the latest breakthroughs in vision research and events in your community by subscribing to our e-newsletter that lands in inboxes the beginning of each month.
